Reprinted with permission from J Clin Aesthet Dermatol. 2023;16(5):31-39
Reprinted with permission from J Clin Aesthet Dermatol. 2023;16(5):31-39
by Linda Stein Gold, MD; Pearl Kwong, MD, PhD; Zoe Draelos, MD;
Krysten Lisella Arekapudi, FNP-C, DNP; Ofra Levy-Hacham, PhD; Maya Erlich, MSc; and Seemal R. Desai, MD
Dr. Stein Gold is with the Department of Dermatology at Henry Ford Health System in Detroit, Michigan. Dr. Kwong is with HCA Healthcare at Mercer University School of Medicine in Orange Park, Florida, and Solutions Through Advanced Research, Inc. in Jacksonville, Florida. Dr. Draelos is with Dermatology Consulting Services in High Point, North Carolina. Ms. Arekapudi is with Prescription Galderma in Dallas, Texas. Dr. Levy-Hacham and Ms. Erlich are with Sol-Gel Technologies in Ness Ziona, Israel. Dr. Desai is with Innovative Dermatology in Plano, Texas, and the University of Texas Southwestern in Dallas, Texas.
FUNDING: Medical writing and editorial support for the preparation of this manuscript was provided by Simpson Healthcare and funded by Galderma Laboratories.
DISCLOSURES: Dr. Stein Gold is an investigator, advisor, and/or speaker for Galderma, Sol Gel, Ortho Dermatologics, Sun Pharma, and Almirall. Dr. Draelos is a researcher and consultant for Galderma. Dr. Desai has served as a consultant for Galderma, and several other industry organizations in a role of a clinical investigator and/or consultant. The remaining authors have no relevant conflicts of interest to disclose related to the content of this article.
ABSTRACT: Objective. Topical therapies remain the mainstay in treating patients with acne and rosacea. However, emerging real-world evidence demonstrates that desired treatment outcomes might not be achieved if patient satisfaction and adherence are low. Poor tolerability of active drug(s) and vehicle components and/or the drug delivery system could negatively influence adherence. Additionally, adherence might be lower with complex treatment regimens involving the application of multiple topical formulations. Optimizing vehicle tolerability and simplifying regimens that use fixed-dose combinations may improve treatment outcomes, better patient satisfaction, and reduce overall treatment costs. This qualitative review discusses several innovative drug delivery technologies and formulations aimed at improving patient satisfaction and adherence.
Methods. The authors conducted a search of current and emerging topical drug delivery technologies used in clinical studies, reviewed primary literature on the chemical characteristics of topical dosage forms, and compared the impacts on treatment outcomes for acne and rosacea.
Results. This article provides insight into innovative vehicles and drug delivery systems that have emerged allowing for fixed-dose combinations of incompatible active drugs and improving the tolerability of historically irritative active ingredients.
Limitations. Further research is needed to fully highlight the impact of patient satisfaction and modern topical formulations on adherence and treatment outcomes.
Conclusion. Drug microencapsulation is a delivery technology that has enabled development of a topical fixed-dose combination of benzoyl peroxide and tretinoin preventing the oxidation of tretinoin by benzoyl peroxide and improving the tolerability of the active ingredients.
Keywords: Vehicles, microencapsulation, technology, tretinoin, benzoyl peroxide, topicals, acne, rosacea
Topical therapy remains a cornerstone treatment approach for a broad range of skin diseases, including acne and rosacea; however, achieving desired outcomes requires patient adherence to the treatment regimen.1 Tolerability of the active drug(s) and components of the delivery vehicle have been shown to increase patient satisfaction and adherence to topical therapies while reducing overall long-term treatment costs.2
Topical retinoids are recommended as first-line treatment for acne vulgaris as monotherapy or in combination with benzoyl peroxide (BPO).3,4 However, unencapsulated combinations of tretinoin and BPO are chemically unstable and can be inactivated when coadministered. Therefore, unencapsulated formulations of tretinoin need to be applied separately from BPO. Additionally, unencapsulated tretinoin can cause photosensitivity.3
Topical treatments that reduce persistent facial erythema and papulopustular lesions are also cornerstones of therapy for patients with rosacea.5 However, patients with rosacea tend to have extremely sensitive skin, and well-tolerated medications, including those with controlled release, are key aspects of successful rosacea management.6
Topical vehicles transfer the required concentration of active drug to the skin’s surface, allowing it to penetrate into the skin and distribute evenly to achieve the desired therapeutic effect.7 For topical treatments requiring skin penetration, the main challenge of drug delivery is transporting bioactive molecule(s) across the stratum corneum (SC) while maintaining the stability of the active drug(s). For a topical treatment to be effective and tolerable, the appropriate concentration of the active drug(s) should remain at the target site for sufficient time to provide a therapeutic effect and limit potential toxicity. Additionally, a vehicle should be soothing, with uniform spreadability and desirable aesthetic attributes to promote patient satisfaction and adherence. These essential attributes of a topical vehicle depend on the selection of suitable components and excipients.7
Poor topical regimen adherence due to drug inefficacy, safety concerns, low tolerability, or regimen complexity has historically caused suboptimal treatment for many skin diseases.8,9 The development and innovation of drug delivery technologies and vehicle composition with improved aesthetic characteristics are essential to achieving treatment success, lowering overall costs, and maintaining patient satisfaction.10
Overview of topical controlled-release technologies used for the treatment of acne and rosacea
Several drug delivery technologies have been developed to administer topical acne and rosacea treatments with improved drug penetration, tolerability, convenience, and overall patient satisfaction and adherence (Table 1). Below is a brief description of several drug delivery technologies.
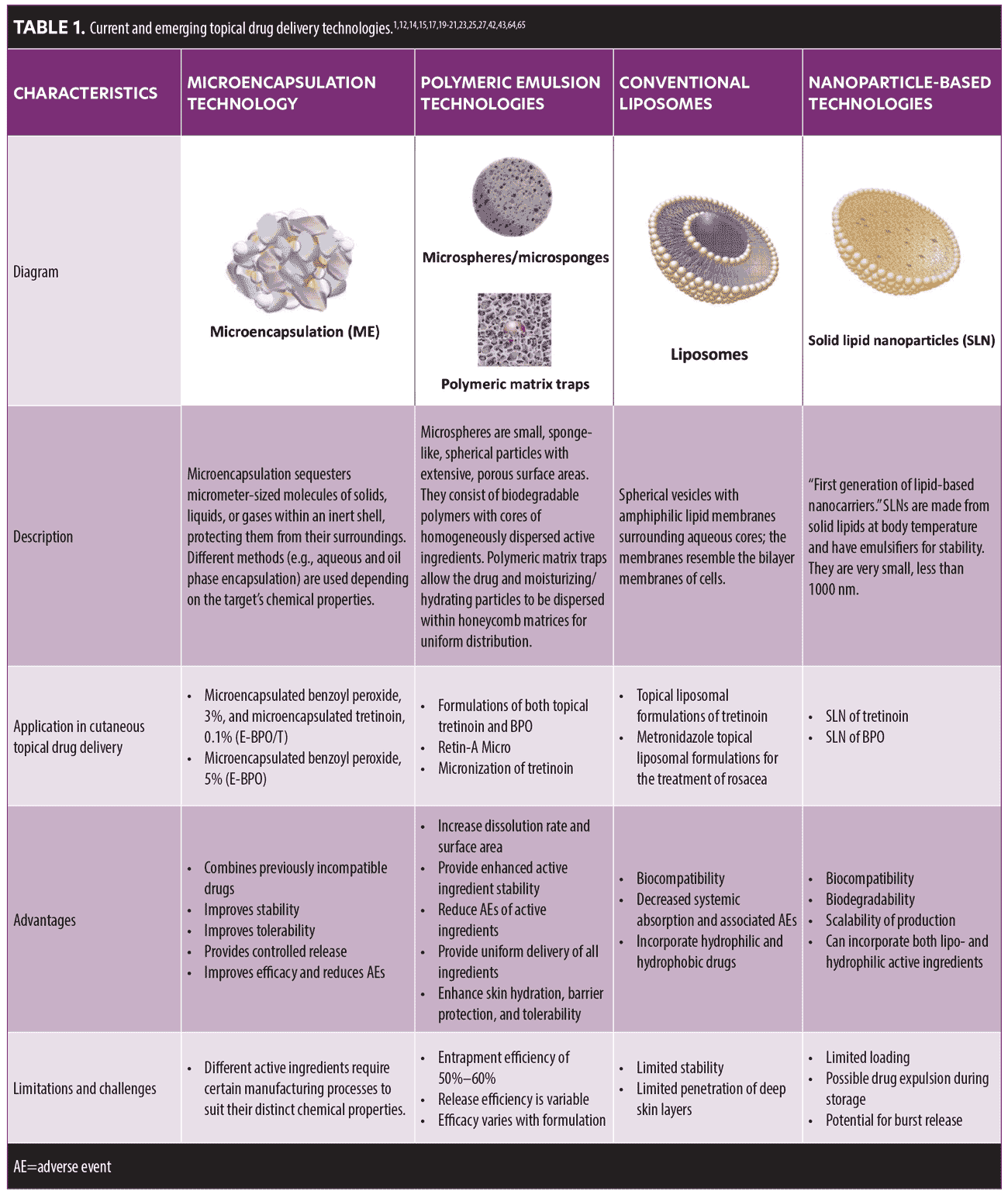
Microencapsulation technology.Microencapsulation (ME) is a process that involves the entrapment of micrometer-sized particles of solids or droplets of liquids or gases in an inert shell that protects the enclosed substances from the surrounding environment.11 Silica microencapsulation is an advanced technology that can be used to enclose active compounds within a silica shell. This process enables the development of controlled-release formulations and the combination of separately encapsulated, historically incompatible active drugs, such as BPO and tretinoin, into fixed-dose formulations.
ME technology via the sol-gel process offers several key advantages over polymeric formulations.11 Amorphous silica is a biologically and chemically inert, highly stable material.12 Thus, silica microencapsulation is suitable for the development of vehicles for a broad range of topical preparations. Moreover, this technology offers several advantages over organic polymer technologies, including the ability to regulate the quantity of the resulting microparticles as well as their shape, density, and surface properties.11
Recently, ME using the sol-gel process was used to develop two new medications in dermatology: a novel 5% microencapsulated BPO (E-BPO) for the treatment of rosacea and a fixed-dose combination of 3% BPO and 0.1% tretinoin (E-BPO/T) approved to treat acne.
Previous evidence supports the efficacy of BPO for the treatment of rosacea; however, BPO has irritative properties that limit its use, especially on hypersensitive rosacea skin.12 ME of BPO crystals within silica shells allows for controlled release onto the skin over time. This measured delivery improves tolerability by avoiding the release of an irritating bolus dose of BPO. Phase III studies of E-BPO have demonstrated efficacy in treating rosacea papules and pustules while maintaining tolerability similar to that of the vehicle control; the adverse events (AEs) were mostly mild to moderate in severity.13
BPO and tretinoin have historically been incompatible within one formulation because tretinoin is highly sensitive to oxidation, and BPO is a strong oxidizer. Thus, such a combination has previously been chemically unstable and had a very short shelf life. In addition, because both active pharmaceutical ingredients are highly irritating to the skin, the unencapsulated combined use of BPO and tretinoin might be intolerable for patients with acne. ME of BPO and tretinoin to form E-BPO/T separates the active ingredients within silica shells, allowing BPO and tretinoin to be combined. ME also acts as a buffer between the active drugs and the skin, providing a slow release of each that improves the tolerability of two historically irritative drugs.12 Phase III trials of E-BPO/T have demonstrated superior efficacy compared with vehicle in treating both inflammatory and noninflammatory lesions of acne and have shown tolerability similar to that of vehicle.
Microsphere-based technologies. Microspheres are tiny, round, biodegradable polymer-based spheres perfused with the active drug.14 These microspheres act as a reservoir system for the active drug, which is dispersed throughout the particle’s core. Following the degradation of the spheres, the active drug is released at the desired time intervals, providing controlled drug release without an increase in transdermal penetration.14 Currently available microsphere formulations consisting of topical BPO and tretinoin have shown enhanced efficacy and improved tolerability in acne treatment.14 However, the entrapment efficiency of microspheres is limited (50%–60%), with a variable release efficiency.15,16
Microsponges are other polymer-based delivery vehicles, composed of rigid sphere structures and porous surfaces that allow them to hold large volumes of the active drug through a meshwork of connecting channels. Studies have shown that microsponges enhance stability, reduce side effects, and allow the controlled release of active drugs onto the skin.17 Microsponges are used for the development of cosmetics and topical products for the treatment of a wide range of skin diseases. Unlike microencapsulation technology, the porous nature of the polymeric microsponges allows the active drugs to diffuse and achieve a concentration equilibrium between the vehicle and the microsphere.14,17 Microsphere-based vehicles, such as tretinoin microsphere gel, 0.04%, have demonstrated efficacy in acne treatment with a low incidence of AEs.18 Additionally, a cream containing microspheres of BPO had favorable efficacy and a very low potential for irritation.14
Polymeric matrix traps. Polymeric matrix traps, also known as polymeric honeycomb matrices, are an emerging strategy for effective drug delivery. The honeycomb matrix structures the active drug with carbomer cross-linked polymers, carbomer copolymer Type B, and homopolymer Type A, evenly distributing the active drug and moisturizing and hydrating components to the skin.19 Polymeric matrix traps are advantageous in their ability to disperse micronized drugs uniformly onto the skin, allowing efficient dispersion into skin crevasses without accumulation of the active drug in single areas on the skin surface.19 In this way, there is a controlled release of active drugs, such as tretinoin, and moisturizing and hydrating ingredients delivered across the skin and into hair follicles, which improves skin hydration and tolerability.20 The matrix remains on the surface of the skin unchanged unless there is a pH or temperature variation or the skin is rubbed.19
Liposomal delivery systems. Liposomes are widely used topical drug delivery systems for pharmaceutical ingredients and cosmetics. Various liposomal topical formulations have been developed to enhance the delivery of bioactive molecules to specific skin layers.21 A liposome is a spherical vesicle composed of a membrane that includes amphiphilic lipids—hydrophilic on one side and lipophilic on the other—enclosing an aqueous core. Liposomes may be used to encase hydrophilic and lipophilic drugs simultaneously because of their amphiphilic nature, enabling the delivery of two types of substances with different properties to the skin to improve therapeutic potential.21 Liposomal formulations containing BPO, clindamycin, and tretinoin have been used for the topical application of several drugs for the treatment of acne and rosacea.14,22
Nanoparticle-based technologies.Several nanoparticle-based drug delivery technologies, including solid lipid nanocarriers (SLNs) and nanostructured lipid carriers (NLCs), have been used to create novel formulations and dosage forms to deliver acne and rosacea treatments, such as tretinoin and antibiotics.21,23 For example, an NLC combining tretinoin and tetracycline was developed to treat acne vulgaris.24 Agar diffusion assays showed high entrapment of tretinoin, ranging from 60 percent to 100 percent, demonstrating the antibacterial activities of the nanosystems against Staphylococcus aureus, Pseudomonas aeruginosa, and Cutibacterium acnes.21,24 Nanoparticle-based formulations, including tretinoin, isotretinoin, and triclosan, have been used to combine agents for the treatment of acne.14 Additionally, a metronidazole-loaded, nanostructured, lipid-based vehicle was shown to improve skin deposition and retention when used to treat patients with rosacea.25 Although nanoparticle-based technologies appear to be promising, there are no successful topical nanoparticle products on the market for acne or rosacea.26
Characteristics of topical dosage forms used for the treatment of acne and rosacea
Topical vehicles are formulated to provide patients with easy-to-use, cosmetically appealing products (Table 2). Ointments and creams are among the most commonly used topical dosage forms for the treatment of a broad range of skin diseases.27 Ointments are typically composed of a hydrophilic greasy base that forms an occlusive layer over the skin, preserving body temperature and preventing water loss. Ointments containing solvents, such as propylene glycol, have been shown to be effective in enhancing percutaneous absorption of topical agents, such as corticosteroids, by improving skin hydration and raising skin temperature.28 Compared with other topical dosage forms, ointments are harder to apply and spread. Additionally, the greasy and sticky nature and feel of ointments can sometimes limit patient satisfaction and adherence. Ointments are not suitable for use in a hot climate, and their properties are not always cosmetically appealing, especially when applied to hair-bearing skin.7 The strong emollient effect of ointments makes them appealing to patients with dry skin conditions.7 Ointments are commonly used on the palms and soles, which respond well to their occlusive properties because of the thickness of the skin.7 Recent evidence has demonstrated the efficacy of ointments as a topical vehicle for the treatment of patients with rosacea. In a recent open-label study, topical preparations of calcineurin inhibitors, such as 0.1% tacrolimus, were investigated in the treatment of rosacea and demonstrated significant improvement of erythema in patients with rosacea subtypes (p<0.05) over 12 weeks.29
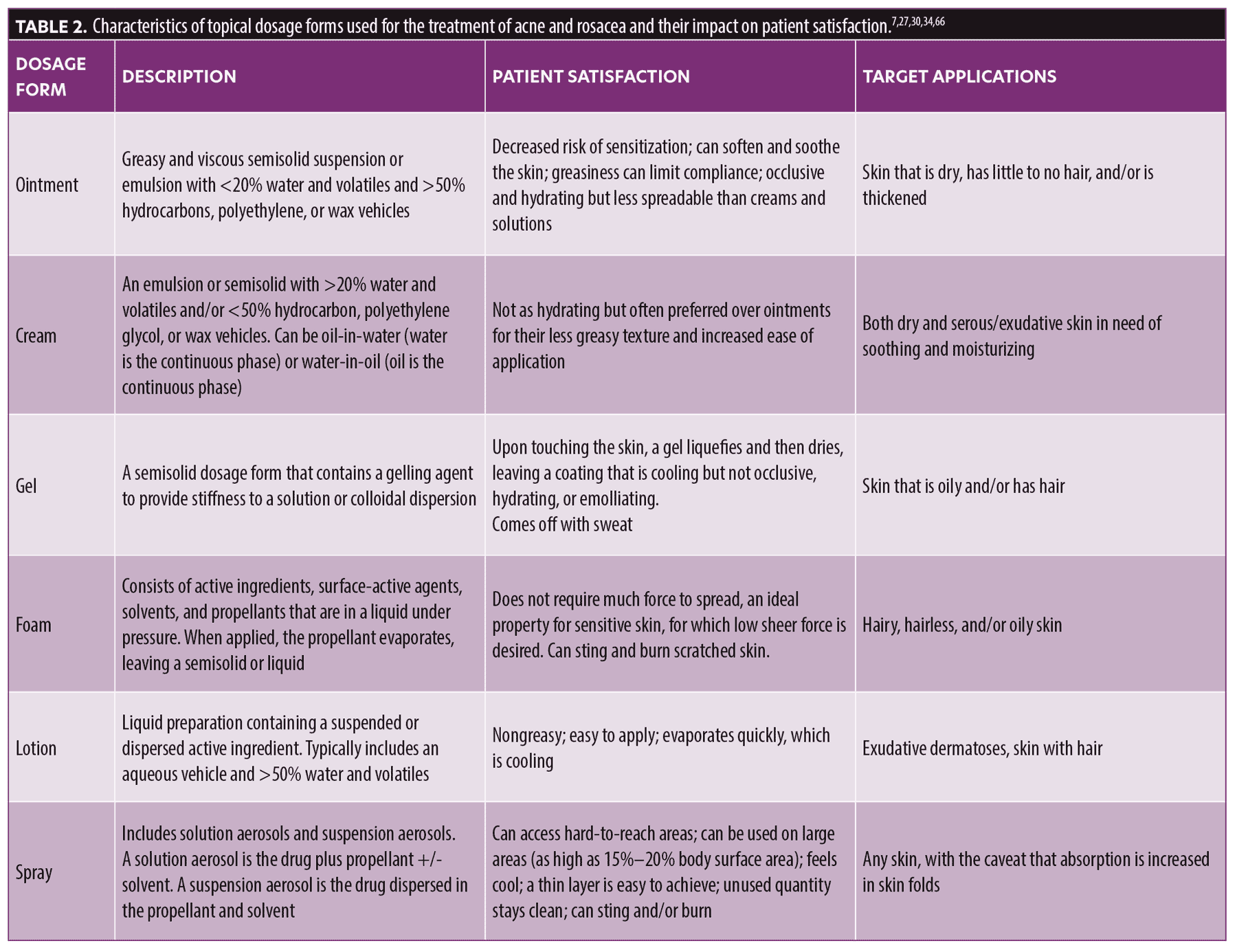
Creams, another commonly used cutaneous topical dosage form, are emulsions of water in oil (oily creams) or oil in water (vanishing creams). The active agent is dispersed between the oil and water phases based on its partition coefficient. Oil-in-water emulsions are best suited for water-soluble drugs, whereas water-in-oil emulsions are best suited for lipid-soluble drugs.7,30 Compared with ointments, creams are significantly less greasy and viscous and are more spreadable and cosmetically appealing.7 Creams also have moistening and emollient properties. Although many patients prefer creams over ointments because of their spreadability and nongreasy nature, creams are comparatively less hydrating because of their limited occlusive properties. Creams have been used to formulate several rosacea treatments, including metronidazole, ivermectin, and azelaic acid.29 In acne, the efficacy and irritation potential of BPO, 4%, cream in a hydro-phase base was investigated in a multicenter, open-label, noncomparative study. By the end of the six-week trial, 85.6 percent of patients experienced a good to very good effect following treatment, and 53.8 percent of patients did not experience any irritation.31
Gels are semisolid systems consisting of suspensions composed of either small inorganic particles (such as clays) or large organic molecules (such as cellulose derivatives and carbomers) interpenetrated by a liquid.32 The three-dimensional network formed within the liquid can be either covalently or physically linked.33 A significant advantage of gels is their cosmetic elegance, which appeals to patients. Furthermore, gels are easy to apply and wash off. However, gels offer no occlusive effects and minimal skin hydration effects. Alcohol-based gels can cause drying of the skin and a stinging sensation.7 Another limitation of gels is that they can be removed by perspiration. Several gel-based formulations have been used to treat rosacea, including azelaic acid, 15%, gel, metronidazole, 0.75%, gel, and brimonidine, 3 mg/g gel.29 Additionally, adapalene 0.1%/BPO 2.5% gel was approved for the treatment of acne. In this combination, drug stability was improved by an aqueous gel vehicle. Moreover, the gel-based combination formulation resulted in greater efficacy than the individually applied drugs.27
Lotions are emulsified liquid dosage forms. They generally contain an aqueous vehicle and contain more than 50 percent water and volatiles that provide a cooling effect upon evaporation.7,34 They are also helpful for application to hairy areas and for the treatment of conditions that affect the scalp. Lotions are easily spread and are useful for large areas because of their ease of application.7 However, lotions are less hydrating than creams and ointments.7,34
Foams are emerging as an attractive topical dosage form with appealing characteristics. Foams are pressurized liquids composed of active ingredients, propellants, surface-active agents, and solvents in an aluminum can with a hydrocarbon propellant (propane/butane), forming a foam lattice of dispersed gas bubbles.24,32 Following valve actuation, a liquid or semisolid product is produced, and the propellant is evaporated.27 Foams offer patients several advantages, including improved skin absorption and low-residue cosmetic appeal. Foams are also less dense and easier to apply and spread on the skin surface. A combination of calcipotriol, 50μg/g, plus betamethasone, 0.5mg/g, in an innovative foam vehicle showed significantly higher steady-state levels of calcipotriol and betamethasone than an ointment formulation of the same concentrations.35 Azelaic acid foam in a 15% concentration has also been shown to be effective and well tolerated in patients with rosacea.29 In acne, foam vehicles offer the advantage over gels by minimizing the absorption of retinoids, thus reducing systemic toxicity and teratogenic potential.27 In a study that compared tazarotene foam (0.1%) versus gel (0.1%), systemic exposure to the retinoid was 35- to 60-fold higher following application of the gel compared with the foam-based formulation.36
Sprays are another emerging topical vehicle. They are composed of droplets of solution with an active drug contained in propellant and combined with a solvent such as ethanol, acetone, hexadecyl alcohol, glycol ethers, or polyglycols.7,32 Sprays can also be formulated in the form of a suspension of insoluble drugs. Key advantages of sprays include their ability to treat large areas of the skin and their easy application, which improve patient adherence.34 However, they may cause stinging and burning sensations upon application.34
Excipients used in topical vehicles
Excipients are pharmaceutical ingredients other than the active component and are added to topical formulations to improve their physicochemical characteristics.37 These include humectants, emollients, emulsifiers and solubilizers, silicone, stiffening/thickening/gelling agents, solvents, penetration enhancers, chelating agents, and antioxidants.38 Certain excipients should be used with caution to avoid aggravating conditions (e.g., while occlusives can treat dry skin, some can also cause acne-like breakouts).39
Humectants are water-soluble substances that can attract water both from within the body (the dermis) as well as from the outside environment. Humectants act by binding water into the SC of the outer epidermis and have been shown to reduce the hyperproliferative reaction to barrier disruption.40 Examples of humectants are lactic acid, glycerin, and glycolic acid. Furthermore, “natural occurring humectants,” such as free amino acids, urea, inorganic salts, and sugars, are packed in corneocytes in the SC layer and play a role in maintaining adequate skin hydration; they are also known as the natural moisturizing factor.39,41 Although humectants can act as emollients, they can conversely increase water loss by pulling water from the dermis to the epidermis, where it then evaporates. To prevent this from happening, occlusive agents can be added to humectant-containing products.39 Humectants can improve acne outcomes when they are used to counter the dryness that treatments like BPO sometimes cause, and both the humectants glycerin and lactic acid are in E-BPO, 5%, cream (for rosacea) and E-BPO/T (for acne).42-44
Emollients improve skin barrier repair and permeability by forming a seal on the skin’s surface.39,40 Long-chain saturated fatty acids, such as stearic, linoleic, oleic, and lauric acids, which are found in various natural oils (e.g., wool fat, palm oil, and coconut oil), are examples of essential fatty acid emollients.7,39 Emollients influence skin physiology by exerting many effects on skin barrier function, including membrane fluidity and the formation of eicosanoids that signal cells and inflammatory pathways.7,40
Emulsifiers are surfactants that allow substances that are not miscible to be present in a single phase. While beneficial in this aspect, some emulsifiers dissolve lipids, resulting in cutaneous irritation. Furthermore, certain nonionic emulsifiers, such as polyethylene glycol, can form peroxides and aldehydes when oxidized. Other examples of emulsifiers are polysorbate 60 and cetyl palmitate.41,45
Silicone comes from silica, a component of granite, quartz, and sand. Due to its strong alternating silica and oxygen bonds, silicone is heat stable and resistant to oxidation.
Furthermore, it is neither soluble nor miscible in water, making it an effective occluding agent that can support the skin barrier and promote the healing of dry and irritated skin. Silicone also acts as an emollient by smoothing the SC and making the skin softer. In addition to these qualities, dermatologic formulations of silicone are also clear, odorless, and nontoxic.44
Stiffening agents, such as paraffin, lanolin, petrolatum, and cetyl alcohol, increase the viscosity of formulations and provide structure to topical semisolids.7,46 Some stiffening agents are also occlusive, providing protection and softening the skin, and are often incorporated into topical vehicles used to treat dry and/or inflammatory skin diseases, such as eczema and psoriasis.7,46 The stiffening agent cetyl alcohol is also found in E-BPO and E-BPO/T, where it can be expected to provide structure and counter any dryness.42,43
Gelling agents give gels most of their structure and increase the viscosity of creams and lotions.34,45 By one suggested definition, a gel is “a semisolid dosage form that contains a gelling agent to provide stiffness to a solution or colloidal dispersion for external application to the skin.”34,45 Agents used for gelling include carbomers, xanthan gum, methyl cellulose, gelatin, and guar gum.45,46 Usually, the gelling agent is dispersed in a hydroalcoholic medium or water to form a consistent dispersion to which the active ingredient and preservative are added to form a medicated gel.46 In theory, the viscosity may impact the delivery and skin retention of the drug dosage form and is an important consideration for topical vehicle design.46
Solvents are incorporated into vehicles to dissolve or disperse the drug. Furthermore, volatile solvents evaporate upon application, leaving what is sometimes called the “secondary formulation” with a different solubility.45 For instance, once the solvent evaporates, the drug may crystallize or dissolve, changing how much of the drug penetrates the skin and how much is retained.46 Examples of solvents are water, propylene glycol, and mineral oil.45
Penetration enhancers are needed to facilitate drug passage through the SC, which is both hydrophobic and dense.47 Properties of the skin that impact penetration include the lipid content of the skin at the place of application, the density of the sweat ducts and hair follicles, body temperature, and skin hydration.47 Fatty acids, such as oleic acid and lauric acid, improve penetration thanks to having a hydrophobicity similar to that of the SC. Alcohols improve penetration by disrupting lipids between the cells of the SC, forming a concentration gradient upon evaporation, and increasing drug solubility in the SC.47 The problem with certain enhancers, such as those that act on lipids, is that they are also irritating.48
Other agents are added to protect ingredients. Chelating agents bolster preservatives by binding to metals and hindering the chemical breakdowns they catalyze. Ethylenediamine tetraacetate is an example of a chelating agent.46 Antioxidants such as butylated hydroxyanisole curtail oxidation, and preservatives such as benzoic acid, methylparaben, and propylparaben curtail the growth of microorganisms.46
Impact of topical vehicles on patient satisfaction, adherence, and treatment outcomes
Tolerability impacts how adherent patients are to treatment, with lack of adherence leading to poor outcomes. Factors that affect tolerability include the skin properties, the vehicle, and the active ingredient. As such, the ingredient characteristics of a pharmaceutical formulation are essential to patient tolerance, adherence, and, ultimately, outcomes.1,49
In a study that included 102 patients with psoriasis, Teixeira and colleagues evaluated the relationship between treatment vehicle and adherence and found that more than 75 percent of patients were nonadherent to treatment.50 Reasons for lack of adherence included forgetfulness, interference with daily activities, and vehicle characteristics, such as staining of clothes, length of time needed for application, and difficulty of spreading. Furthermore, the researchers found that when a large body area was treated, adherence was greater with gels and creams versus ointments, but the inverse was true with small areas.50
In a study that included 500 patients with acne, adherence to treatment was found to be poor in 64 percent of patients, in part due to dissatisfaction with treatment. The use of oral isotretinoin and satisfaction with treatment were identified as independent factors that affected patient adherence. Of the patients included in the study, 51.8 percent were satisfied with their treatments, and treatment satisfaction was higher among patients treated with oral isotretinoin.51
Optimized formulations with ingredients that provide moisturization, such as humectants and emollients, reduce dryness and irritation in patients with acne, thereby improving tolerability and, potentially, adherence. In a split-face acne study involving 43 participants with acne, participants applied tretinoin cream, 0.025%, to both sides of the face, and then clindamycin-BPO to one-half of the face and clindamycin-BPO with an “optimized vehicle” (glycerin and dimethicone) to the other half. At various time points, the areas treated with the optimized vehicle displayed significantly (p<0.05) less erythema, dryness, and peeling, and participants reported significantly less dryness and burning.44
Additionally, the AEs associated with topical cutaneous drugs or specific formulations can result in a high rate of treatment discontinuation, affecting the quality and cost of care.52 To determine how AEs impacted treatment discontinuation and healthcare costs in patients with rosacea treated with topical therapies, Williamson and colleagues completed a retrospective cohort study.52 Ultimately, they evaluated 49,351 patients, most of whom were female (74.5%) and middle-aged (mean 54 years old).52 They found that of the 6270 patients with a coded AE, only 3.2 percent continued the treatment. Instead, most (89.4%) stopped treatment within a month or so (31 days), and 7.4 percent changed treatments within nine days. Furthermore, average rosacea-related costs were significantly (p<0.001) higher for those with an AE than for those without ($325 versus $172, respectively). Furthermore, of those treatments associated with AEs, the highest percentage of patients who experienced an AE were those treated with BPO (22.5%) versus 12.3 to 14.3 percent for azelaic acid, metronidazole, and sodium sulfacetamide/sulfur. The authors concluded that the AEs affected treatment quality and cost and that different therapies, or those with new formulations, may improve adherence and symptoms.52
Research highlights how convenience (combined treatments) is associated with increased adherence and improved outcomes. In a study that compared a clindamycin phosphate, 1.2%, and tretinoin, 0.025%, gel combination product with separate daily applications of clindamycin phosphate gel, 1%, and tretinoin cream, 0.025%, in patients with mild to moderate acne vulgaris, the median adherence to the combination product was 88 percent compared with 61 percent for the products applied separately.53 There was a 51 percent mean reduction in total lesions for the combination gel group versus a 32 percent mean reduction for the clindamycin phosphate, 1%, plus tretinoin, 0.025%, gel group by the end of the study.53
Emerging novel vehicles and technologies for the treatment of acne and rosacea and their impact on patient satisfaction
Patient expectations and satisfaction with their acne and rosacea therapy are important aspects of disease management. Improved adherence and patient outcomes, including quality of life (QoL) measures, correlate with medications that patients perceive to be safe, effective, simple to use, and convenient.51,54
Fixed-dose formulations with increased tolerability have been shown to improve patient satisfaction and QoL, and several innovative fixed-combination topical treatments have been developed.55 The recently approved E-BPO/T cream, indicated for acne vulgaris, was formulated by combining encapsulated BPO and encapsulated tretinoin with humectants, a chelating agent, emulsifiers, an emollient, a thickening agent, and a gelling agent.43 Microencapsulation enhances the properties of the vitamin A derivative, tretinoin, improves compatibility and tolerability of BPO and tretinoin in a single vehicle, and provides a slower release of both agents to the skin.22
The advantages of combining BPO and tretinoin in a single formulation are multiple. Both BPO and topical retinoids are recommended first-line acne treatments.3 BPO can prevent bacterial resistance and should be incorporated into both topical and systemic antibiotic acne regimens.3 Tretinoin is an effective acne treatment because it is anti-inflammatory, resolves microcomedone precursors, and lyses comedones.3 Furthermore, skin clearance achieved with oral therapies can be maintained with tretinoin.3
However, traditional BPO and tretinoin also have limitations. Both BPO and tretinoin can be irritating, with tretinoin potentially causing skin dryness, peeling, and redness.3 In earlier formulations, tretinoin was solubilized in alcohol or myristate. Although this allowed for rapid tretinoin release, these formulations could be irritating.49 Traditional tretinoin is also unstable in light.56 Furthermore, while traditional BPO and tretinoin can be used together in treatment, traditional BPO can oxidize a retinoid when given simultaneously, necessitating application at different times (e.g., morning and evening), sacrificing convenience.57 By encapsulating BPO and tretinoin in separate microcapsules, skin irritation is reduced, and oxidation of tretinoin by BPO is limited.12 These actions make the combination both more tolerable and more stable.12
Other BPO combinations have also been formulated, including BPO and clindamycin in an aqueous gel. The rationale behind developing an aqueous combination is that both BPO and clindamycin effectively treat acne, and the combination is superior to either given alone. Still, BPO can be difficult for some patients to tolerate, limiting its use. Furthermore, the combination enables the treatments to be given once a day, which may improve adherence.58
In two identical randomized, double-blind, active- and vehicle-controlled studies that compared clindamycin phosphate 1.2%/BPO 2.5% gel with the individual ingredients—clindamycin phosphate, benzoyl peroxide, and vehicle—in people 12 years and older with moderate to severe acne, researchers found that treatment with the gel resulted in significantly (p<0.001 to p=0.002) better reductions in acne severity and inflammatory and noninflammatory lesions compared with its individual ingredients.58
Improvement in health-related QoL with clindamycin phosphate 1.2%/BPO 2.5% gel was subsequently evaluated in a large acne QoL study.59 The study demonstrated that patients’ health-related QoL improved significantly (p<0.001) with clindamycin phosphate 1.2%/BPO 2.5% gel treatment compared with the individual ingredients and vehicle in moderate to severe acne across all four of the assessed QoL domains: acne symptoms, role-emotional, self-perception, and role-social.59
Tretinoin is also combined with other active ingredients to capitalize on complementary mechanisms of action.1,60 For instance, tretinoin prevents the formation of comedones and facilitates their expulsion, while clindamycin decreases inflammatory lesions and inhibits bacteria.1 To combine these ingredients, scientists developed an aqueous-based gel with tretinoin, 0.025%, and clindamycin phosphate, 1.2%. The formulation solubilizes and stabilizes the active ingredients and allows them to be administered once a day.1 Furthermore, solubilizing tretinoin may reduce irritation and improve tolerability via slower cutaneous delivery.60
In a multicenter, randomized, active-drug, vehicle-controlled study that compared tretinoin, 0.025%, and clindamycin phosphate, 1.2%, in an aqueous-based gel to its components and vehicle alone, the efficacy of the combination was superior to treatment with the individual components.61 Moreover, the tolerability of the combination appeared to be as good as that of tretinoin alone, again underscoring the importance of the vehicle to outcomes.61
Given that tretinoin can be irritating, researchers compared the safety and efficacy of tretinoin microsphere gel, 0.04%, with the 0.1% concentration. Of 156 patients, half randomized to each group, there were no significant differences between groups in change in lesion count from baseline to Weeks 2, 4, 8, and 12. Still, the reduction in inflammatory lesions was significantly (p<0.048) greater at Week 2 for the 0.1% group. Interestingly, while there were no significant differences between groups for peeling, burning/stinging, or itching, the proportion of patients with worsening erythema at Week 12 was significantly (p=0.035) higher in the 0.04% group, while significantly more patients experienced dryness at Weeks 2 and 4 in the 0.1% group. These results suggest that higher concentrations in novel formulations can be tolerated, with the additional benefit of bringing about clinical improvement earlier.62
Another study of a novel tretinoin formulation involved a post-hoc analysis of two identical, double-blind, multicenter studies in which patients with moderate to severe acne were treated with tretinoin, 0.05%, lotion (created using micronization and polymeric emulsion) or vehicle applied once a day to the face for 12 weeks.63 In this post hoc analysis, the researchers evaluated safety (erythema and scaling), tolerability (itching, burning/stinging), and both hyperpigmentation and hypopigmentation.62 The most common treatment-related AEs were application site pain, dryness, and erythema, which occurred in 1.4 to 3.7 percent of the tretinoin, 0.05%, group and significantly more (p<0.001) than in the vehicle group, but less than reported in studies of other formulations (tretinoin microspheres and gel).3 Furthermore, erythema and itching improved from baseline to Week 12 in the tretinoin group, and scaling, burning, and stinging only temporarily increased a small amount from baseline to Week 4.62 Postinflammatory hyperpigmentation did not worsen. These findings underscore the benefits of novel drug formulations on safety and tolerability.62
Conclusion
Advances in topical vehicle formulations have enabled the development of novel, patient-centered, fixed-dose combination therapies for acne, rosacea, and other skin disorders. Improved topical vehicles can simplify treatment and improve tolerability, and they are expected to continue improving patient satisfaction, adherence, and treatment outcomes. Drug microencapsulation technology using the sol-gel process has paved the way for innovative fixed-dose combined formulations of drugs that were historically incompatible, advancing the treatment of patients with rosacea and acne.
References
- Hoffman LK, Bhatia N, Zeichner J, et al. Topical vehicle formulations in the treatment of acne. J Drugs Dermatol. 2018;17(6):s6–s10.
- Kircik LH. Importance of vehicles in acne therapy. J Drugs Dermatol. 2011;10(6):s17–23.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74(5):945–973.e33.
- Leyden J, Stein-Gold L, Weiss J. Why topical retinoids are mainstay of therapy for acne. Dermatol Ther (Heidelb). 2017;7(3):293–304.
- Del Rosso JQ, Thiboutot D, Gallo R, et al. Consensus recommendations from the American Acne & Rosacea Society on the management of rosacea, part 5: a guide on the management of rosacea. Cutis. 2014;93(3):134–138.
- Jackson JM, Pelle M. Topical rosacea therapy: the importance of vehicles for efficacy, tolerability and compliance. J Drugs Dermatol. 2011;10(6):627–633.
- Barnes TM, Mijaljica D, Townley JP, et al. Vehicles for drug delivery and cosmetic moisturizers: review and comparison. Pharmaceutics. 2021;13(12):2012.
- Daudén E, Bewley A, Lambert J, et al. Expert recommendations: the use of the fixed combination calcipotriol and betamethasone dipropionate gel for the topical treatment of psoriasis. J Eur Acad Dermatol Venereol. 2014;28(suppl 2):22–32.
- Reich K, Daudén E. Treatment adherence: a hurdle for real-life effectiveness in psoriasis? J Eur Acad Dermatol Venereol. 2014;28(suppl 2):1–3.
- Bewley A, Page B. Maximizing patient adherence for optimal outcomes in psoriasis. J Eur Acad Dermatol Venereol. 2011;25(suppl 4):9–14.
- Ciriminna R, Sciortino M, Alonzo G, et al. From molecules to systems: sol-gel microencapsulation in silica-based materials. Chem Rev. 2011;111(2):765–789.
- Erlich M, Arie T, Koifman N, et al. Structure elucidation of silica-based core-shell microencapsulated drugs for topical applications by cryogenic scanning electron microscopy. J Colloid Interface Sci. 2020;579:778–785.
- Goldgar C, Keahey DJ, Houchins J. Treatment options for acne rosacea. Am Fam Physician. 2009;80(5):461–468.
- Vyas A, Kumar Sonker A, Gidwani B. Carrier-based drug delivery system for treatment of acne. ScientificWorldJournal. 2014;2014:276260.
- Kircik LH. Microsphere technology: hype or help? J Clin Aesthet Dermatol. 2011;4(5):27–31.
- Chadawar V, Shaji J. Microsponge delivery system. Curr Drug Deliv. 2007;4(2):123–129.
- Kaity S, Maiti S, Ghosh AK, et al. Microsponges: a novel strategy for drug delivery system. J Adv Pharm Technol Res. 2010;1(3):283–290.
- Dogra S, Sumathy TK, Nayak C, et al. Efficacy and safety comparison of combination of 0.04% tretinoin microspheres plus 1% clindamycin versus their monotherapy in patients with acne vulgaris: a phase 3, randomized, double-blind study. J Dermatol Treat. 2021;32(8):925–933.
- Kircik LH, Draelos ZD, Berson DS. Polymeric emulsion technology applied to tretinoin. J Drugs Dermatol. 2019;18(4):s148–s154.
- Baldwin H, Webster G, Stein Gold L, et al. 50 years of topical retinoids for acne: evolution of treatment. Am J Clin Dermatol. 2021;22(3):315–327.
- Garg T. Current nanotechnological approaches for an effective delivery of bio-active drug molecules in the treatment of acne. Artif Cells Nanomed Biotechnol. 2016;44(1):98–105.
- Green LJ, Lain E, Prunty T, et al. Enhancing topical pharmacotherapy for acne and rosacea: vehicle choices and outcomes. J Clin Aesthet Dermatol. 2022;15(5):36–40.
- Müller RH, Radtke M, Wissing SA. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002;54(suppl 1):S131–S155.
- Lin CH, Fang YP, Al-Suwayeh SA, et al. Percutaneous absorption and antibacterial activities of lipid nanocarriers loaded with dual drugs for acne treatment. Biol Pharm Bull. 2013;36(2):276–286.
- Shinde UA, Parmar SJ, Easwaran S. Metronidazole-loaded nanostructured lipid carriers to improve skin deposition and retention in the treatment of rosacea. Drug Dev Ind Pharm. 2019;45(7):1039–1051.
- Chutoprapat R, Kopongpanich P, Chan LW. A mini-review on solid lipid nanoparticles and nanostructured lipid carriers: topical delivery of phytochemicals for the treatment of acne vulgaris. Molecules. 2022;27(11):3460.
- Rosen J, Landriscina A, Friedman AJ. Principles and approaches for optimizing therapy with unique topical vehicles. J Drugs Dermatol. 2014;13(12):1431–1435.
- Dhar S, Seth J, Parikh D. Systemic side-effects of topical corticosteroids. Indian J Dermatol. 2014;59(5):460–464.
- Schaller M, Schöfer H, Homey B, et al. Rosacea management: update on general measures and topical treatment options. J Dtsch Dermatol Ges. 2016;14(suppl 6):17–27.
- Buhse L, Kolinski R, Westenberger B, et al. Topical drug classification. Int J Pharm. 2005;295(1-2):101–112.
- Sawleshwarkar SN, Salgaonkar V, Oberai CM. Multicenter study to evaluate efficacy and irritation potential of benzoyl peroxide 4% cream in hydrophase base (Brevoxyl) in acne vulgaris. Indian J Dermatol Venereal Leprol. 2003;69(1):19–22.
- Topical and transdermal drug products. Pharmacopeial Forum. 2009;35(3):750–764.
- United States Pharmacopeial Convention. (1151) Pharmaceutical Dosage Forms. May 2021. https://latam-edu.usp.org/wp-content/uploads/2021/08/1151.pdf. Accessed May 14, 2022.
- Mayba JN, Gooderham MJ. A guide to topical vehicle formulations. J Cutan Med Surg. 2018;22(2):207–212.
- Tada Y, Iversen L, Koo J. Early efficacy and safety data with fixed-dose combination calcipotriol/betamethasone dipropionate foam attributed to mechanism of absorption and steroid potency. J Eur Acad Dermatol Venereol. 2021;35(suppl 1):5–9.
- Jarratt M, Werner CP, Alió Saenz AB. Tazarotene foam versus tazarotene gel: a randomized relative bioavailability study in acne vulgaris. Clin Drug Investig. 2013;33(4):283–289.
- Caballero ML, Quirce S. Immediate hypersensitivity reactions caused by drug excipients: a literature review. J Investig Allergol Clin Immunol. 2020;30(2):86–100.
- Kalasz H, Antal I. Drug excipients. Curr Med Chem. 2006;13:2535–2563.
- Purnamawati S, Indrastuti N, Danarti R, et al. The role of moisturizers in addressing various kinds of dermatitis: a review. Clin Med Res. 2017;15(3–4):75–87.
- Proksch E, Berardesca E, Misery L, et al. Dry skin management: practical approach in light of latest research on skin structure and function. J Dermatol Treat. 2020;31(7):716–722.
- Lodén M. Role of topical emollients and moisturizers in the treatment of dry skin barrier disorders. Am J Clin Dermatol. 2003;4(11):771–788.
- Galderma Laboratories, L.P. Epsolay (benzoyl peroxide) prescribing information. Updated April 2022. https://www.galderma.com/us/sites/default/files/2022-03/Epsolay_PI.pdf. Accessed June 20, 2022.
- Galderma Laboratories, L.P. Twyneo (tretinoin and benzoyl peroxide) prescribing information. Updated July 2021. https://www.galderma.com/us/sites/default/files/2022-02/Twyneo_PI.pdf. Accessed June 20, 2022.
- Draelos ZD, Callender V, Young C, et al. The effect of vehicle formulation on acne medication tolerability. Cutis. 2008;82(4):281–284.
- Danby SG, Draelos ZD, Gold LFS, et al. Vehicles for atopic dermatitis therapies: more than just a placebo. J Dermatol Treat. 2022;33(2):685–698.
- Chang RK, Raw A, Lionberger R, et al. Generic development of topical dermatologic products: formulation development, process development, and testing of topical dermatologic products. AAPS J. 2013;15(1):41–52.
- Ramadon D, McCrudden MTC, Courtenay AJ, et al. Enhancement strategies for transdermal drug delivery systems: current trends and applications. Drug Deliv Transl Res. 2022;12(4):758–791.
- Benson HA. Transdermal drug delivery: penetration enhancement techniques. Curr Drug Deliv. 2005;2(1):23–33.
- Kircik LH. Evaluating tretinoin formulations in the treatment of acne. J Drugs Dermatol. 2014;13(4):466–470.
- Teixeira A, Teixeira M, Almeida V, et al. Does the vehicle matter? Real-world evidence on adherence to topical treatment in psoriasis. Pharmaceutics. 2021;13(10):1539.
- Hayran Y, İncel Uysal P, Öktem A, et al. Factors affecting adherence and patient satisfaction with treatment: a cross-sectional study of 500 patients with acne vulgaris. J Dermatol Treat. 2021;32(1):64–69.
- Williamson T, Kamalakar R, Ogbonnaya A, et al. Rate of adverse events and healthcare costs associated with the topical treatment of rosacea. Am Health Drug Benefits. 2017;10(3):113–119.
- Yentzer BA, Ade RA, Fountain JM, et al. Simplifying regimens promotes greater adherence and outcomes with topical acne medications: a randomized controlled trial. Cutis. 2010;86(2):103–108.
- Gold MH. Clindamycin phosphate 1.2% and benzoyl peroxide 2.5% gel for the treatment of moderate-to-severe acne: an update. J Clin Aesthet Dermatol. 2012;5(1):30–35.
- Williamson T, Cheng WY, McCormick N, et al. Patient preferences and therapeutic satisfaction with topical agents for rosacea: a survey-based study. Am Health Drug Benefits. 2018;11(2):97–106.
- Martin B, Meunier C, Montels D, et al. Chemical stability of adapalene and tretinoin when combined with benzoyl peroxide in presence and in absence of visible light and ultraviolet radiation. Br J Dermatol. 1998;139(suppl 52):8–11.
- Yang Z, Zhang Y, Lazic Mosler E, et al. Topical benzoyl peroxide for acne. Cochrane Database Syst Rev. 2020;3(3):CD011154.
- Thiboutot D, Zaenglein A, Weiss J, et al. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 2.5% for the once-daily treatment of moderate to severe acne vulgaris: assessment of efficacy and safety in 2813 patients. J Am Acad Dermatol. 2008;59(5):792–800.
- Webster G, Thiboutot DM, Chen DM, et al. Impact of a fixed combination of clindamycin phosphate 1.2%-benzoyl peroxide 2.5% aqueous gel on health-related quality of life in moderate to severe acne vulgaris. Cutis. 2010;86(5):263–267.
- Schlessinger J, Menter A, Gold M, et al. Clinical safety and efficacy studies of a novel formulation combining 1.2% clindamycin phosphate and 0.025% tretinoin for the treatment of acne vulgaris. J Drugs Dermatol. 2007;6(6):607–615.
- Jarratt MT, Brundage T. Efficacy and safety of clindamycin-tretinoin gel versus clindamycin or tretinoin alone in acne vulgaris: a randomized, double-blind, vehicle-controlled study. J Drugs Dermatol. 2012;11(3):318–326.
- Berger R, Barba A, Fleischer A, et al. A double-blinded, randomized, vehicle-controlled, multicenter, parallel-group study to assess the safety and efficacy of tretinoin gel microsphere 0.04% in the treatment of acne vulgaris in adults. Cutis. 2007;80(2):152–157.
- Harper JC, Roberts WE, Zeichner JA, et al. Novel tretinoin 0.05% lotion for the once-daily treatment of moderate-to-severe acne vulgaris: assessment of safety and tolerability in subgroups. J Dermatol Treat. 2020;31(2):160–167.
- Ghasemiyeh P, Mohammadi-Samani S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: applications, advantages and disadvantages. Res Pharm Sci. 2018;13(4):288–303.
- Pokharkar VB, Mendiratta C, Kyadarkunte AY, et al. Skin delivery aspects of benzoyl peroxide-loaded solid lipid nanoparticles for acne treatment. Ther Deliv. 2014;5(6):635–652.
- Weiss SC. Conventional topical delivery systems. Dermatol Ther. 2011;24(5):471–476.